Decades of scientific work allowed the development and production in record time of vaccines against COVID-19.
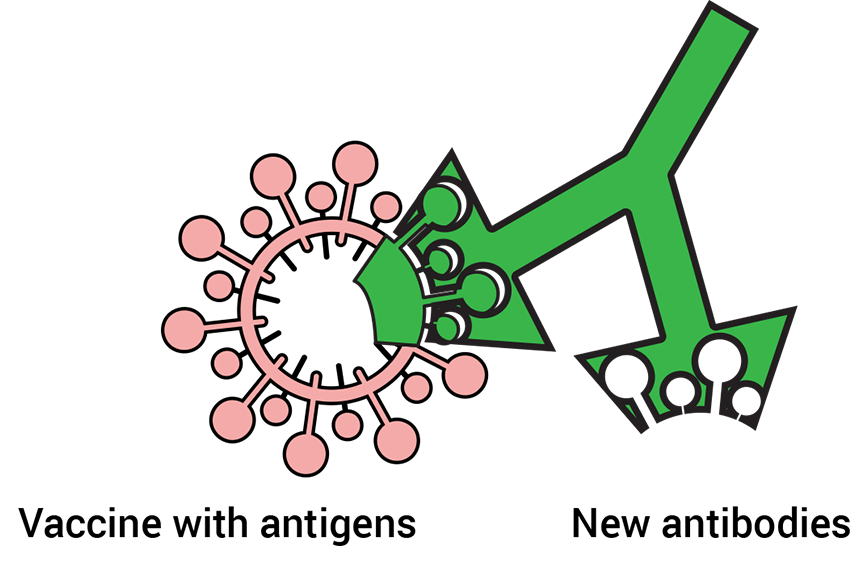
When someone is vaccinated, they will most likely be protected against the target disease. A small number of people cannot be vaccinated – for example, transplant patients, the immunosuppressed, some chronically ill, and also new-borns.
The reason why these people cannot be vaccinated is related to the functioning of their immune system, or because they may develop severe allergies to some of the fluids used as a base to dissolve the vaccine, the excipients.
However, vaccination not only protects those who are vaccinated, but also those who cannot receive the vaccine. When much of the population is already immune, the infectious agent has difficulty spreading in the community and the likelihood of infecting people who cannot be vaccinated is then more limited. This phenomenon is called herd immunity.
What is herd immunity?
i3S - Instituto de Investigação e Inovação em Saúde (Institute for Research and Innovation in Health) April 2020

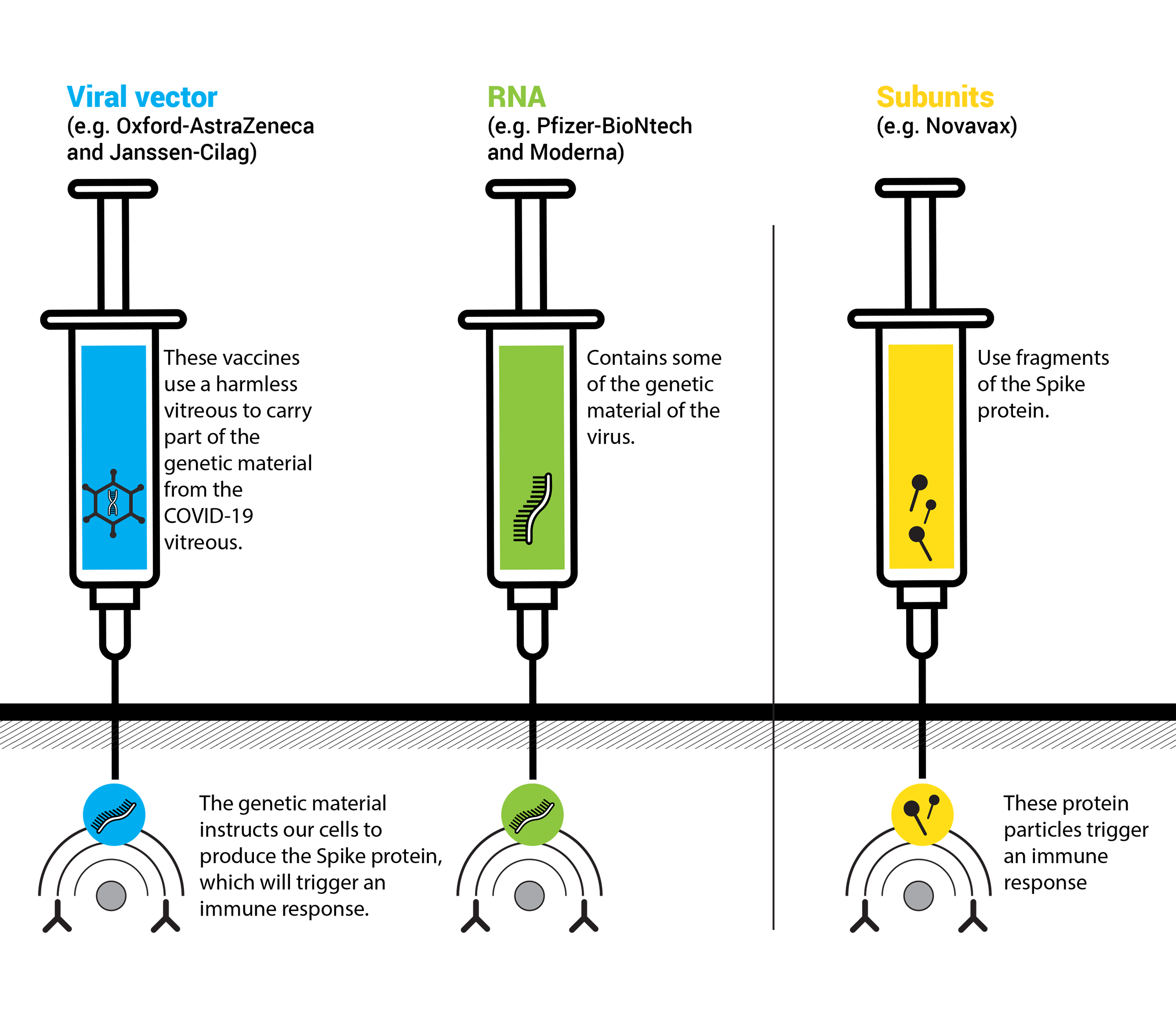
Although all available vaccines have been designed for this target, their development was based on different technologies. There are three main groups of COVID-19 vaccines, two already approved – Viral Vector and RNA – and one that is close to approval - Subunits.
Viral vector: these vaccines use another modified virus – adenovirus – as a vehicle to carry into cells the genetic instructions that will lead to the production of the 'Spike' protein.
RNA (ribonucleic acid): these vaccines use the genetic material of the virus which has the instructions to make the “Spike” protein.
Subunits: these vaccines the “Spike” protein
Ciência Viva, with Paula Alves (IBET e ITQB Universidade Nova de Lisboa) - January 2021



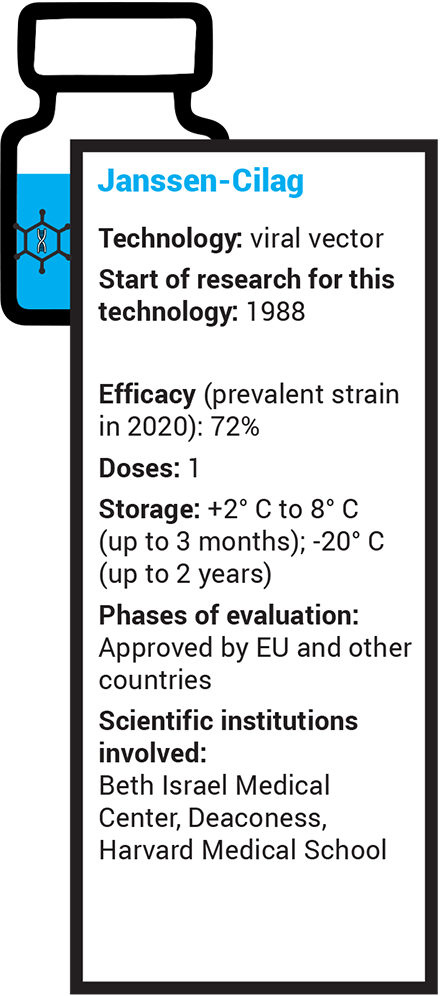
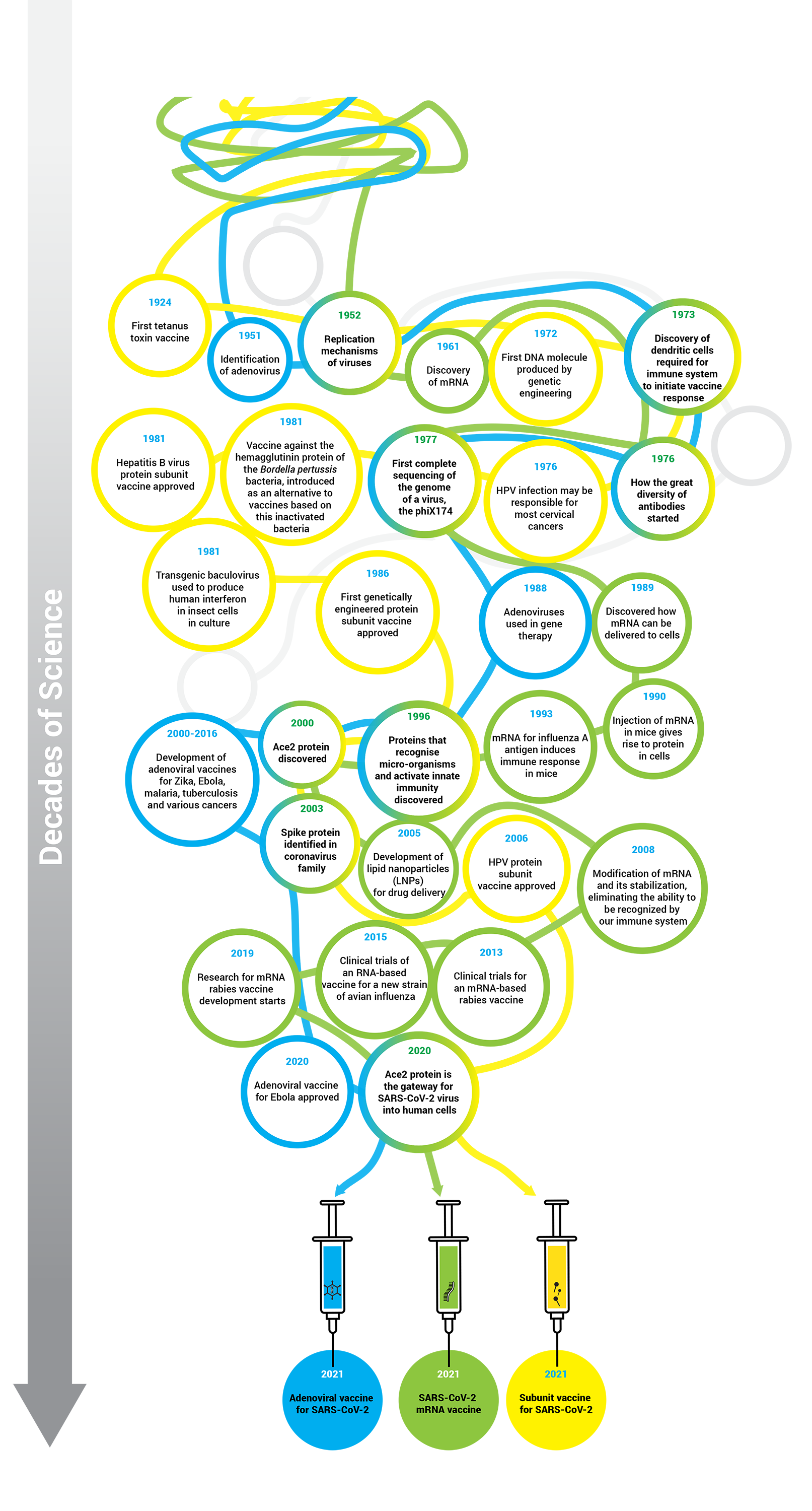
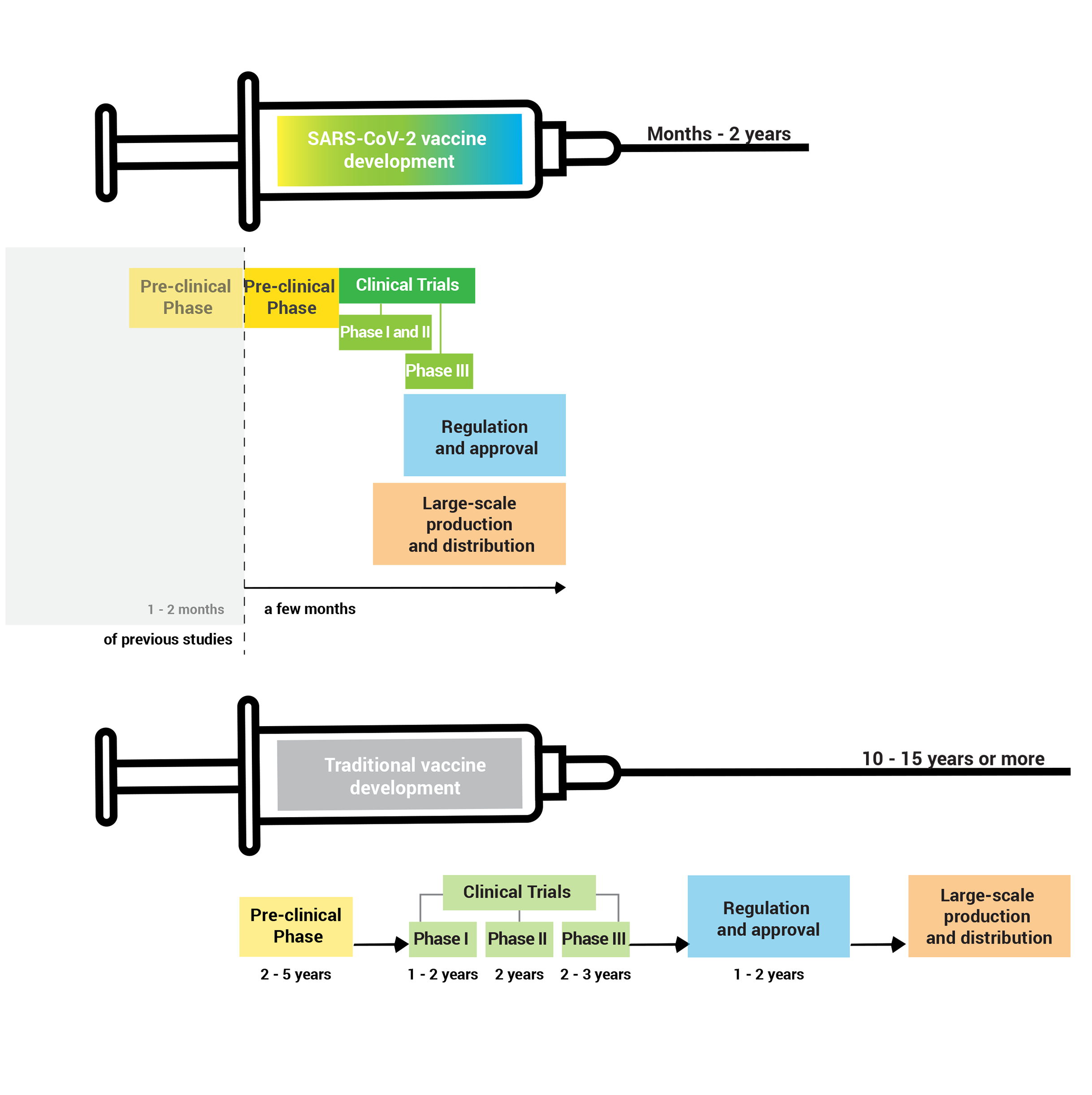
iMM - Instituto de Medicina Molecular João Lobo Antunes (Institute of Molecular Medicine João Lobo Antunes), with Bruno Silva Santos, iMM immunologist - january 2021
iMM - Instituto de Medicina Molecular João Lobo Antunes (Institute of Molecular Medicine João Lobo Antunes), with Miguel Prudêncio, iMM parasitologist - january 2021